SVS Management of Diabetic Foot Guidelines
Key Points
- Diabetes is one of the leading causes of chronic disease and limb loss worldwide, currently affecting 382 million people
- Every year, >1 million people with diabetes suffer limb loss as a result of diabetes.
- Approximately 80% of diabetes-related lower extremity amputations are preceded by a foot ulcer.
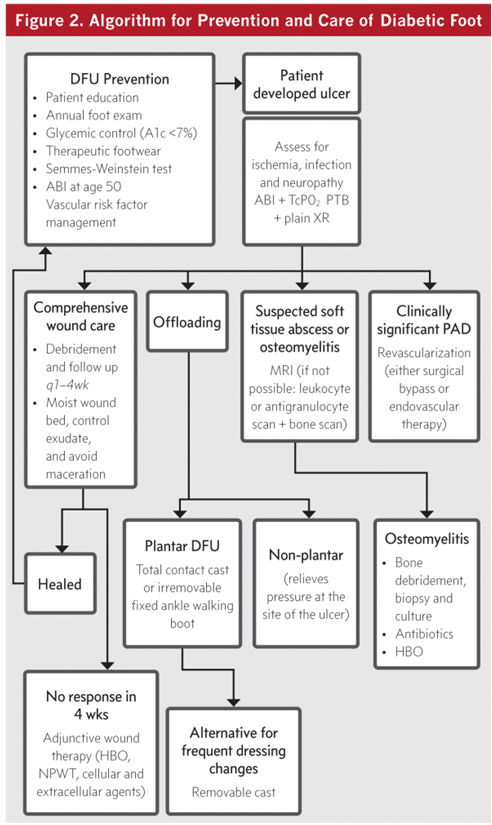
- This progression from foot ulcer to amputation lends to several possible steps where intervention based on evidence- based guidelines may prevent major amputation.
Prevention
Prevention of Diabetic Foot Ulceration
- We recommend that patients with diabetes undergo annual interval foot inspections by physicians (MD, DO, DPM) or advanced practice providers with training in foot care.
- We recommend that foot examination include testing for peripheral nuropathy using the Semmes- Weinstein test.
- We recommend education of the patients and their families about preventive foot care.
Therapeutic Footwear:
- We suggest against the routine use of specialised therapeutic footwear in average risk diabetic patients.
- We recommend using custom therapeutic footwear in high risk diabetic patients, including those with significant neuropathy, foot deformities, or previous amputation.
- We suggest adequate glycemic control (haemoglobin A1c <7% with strategies to minimize hypoglycaemia) to reduce the incidence of diabetic foot ulcers (DFUs) and infections, with subsequent risk of amputation.
- We recommend against prophylactic arterial revascularization to prevent DFU.

Diagnosis of Diabetic Foot Osteomyelitis (DFO)
- In patients with a diabetic foot Infection (DFI) with an open wound, we suggest doing a probe to bone (PTB) test to aid in diagnosis.
- In all patients presenting with a new DFI, we suggest that serial plain radiographs of the affected foot be obtained to identify bone abnormalities (deformity, destruction) as well as soft tissue gas and radiopaque foreign bodies.
- For those patients who require additional (i.e. more sensitive or specific) imaging, particularly when soft tissue abscess is suspected or the diagnosis of osteomyelitis remains uncertain, we recommend using Magnetic Resonance Imaging (MRI) as the study of choice.
- MRI is a valuable tool for diagnosis of osteomyelitis if PTB test is inconclusive or if the plain film is not useful.
- In patients with a suspected DFO for whom MRI is contraindicated or unavailable, we suggest a leukocyte or antigranulocyte scan, preferably combined with a bone scan as the best alternative.
- In patients at high risk for DFO, we recommend that the diagnosis is most definitely established by the combined findings on the bone culture and histology. When bone is debrided to treat osteomyelitis, we recommend sending a sample for culture and histology.
- For patients not undergoing bone debridement, we suggest that clinicians consider obtaining a diagnostic bone biopsy when faced with diagnostic uncertainty, inadequate culture information, or failure of response to empirical treatment.
Assessment
Table 1: Suggested frequency for Follow-UP Evaluation
Peripheral Arterial Disease (PVD) and the DFU
- We suggest that patients with diabetes have ankle- brachial index (ABI) measurements performed when they have reached 50 years of age.
- We suggest that patients with diabetes who have a prior history of DFU, prior intervention for peripheral vascular disease, or known atherosclerotic cardiovascular disease (eg. Coronary, cerebra, or renal) have an annual vascular examination of the lower extremities and feet including ABI and toe pressures.
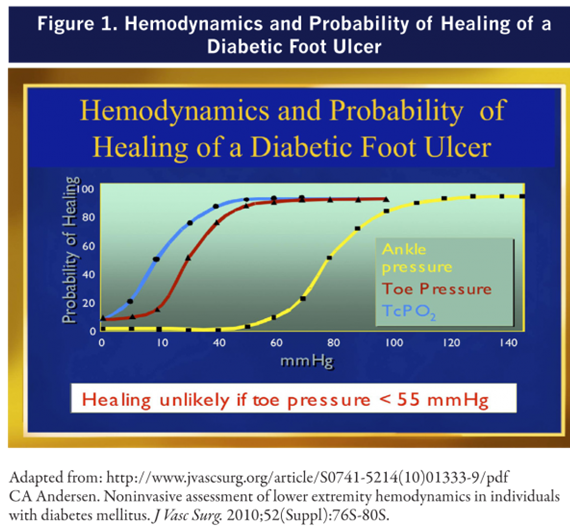
- We recommend that patients with DFU have pedal perfusion assessed by ABI, ankle and pedal Doppler arterial waveforms, and either toe systolic pressure or transcutaneous oxygen pressure (TcPO2)
- In patients with DFU who have PAD, we recommend revascularization by either surgical bypass or endovascular therapy.
Technical and implementation Remarks
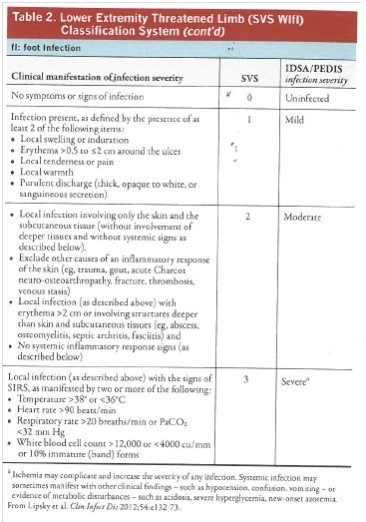
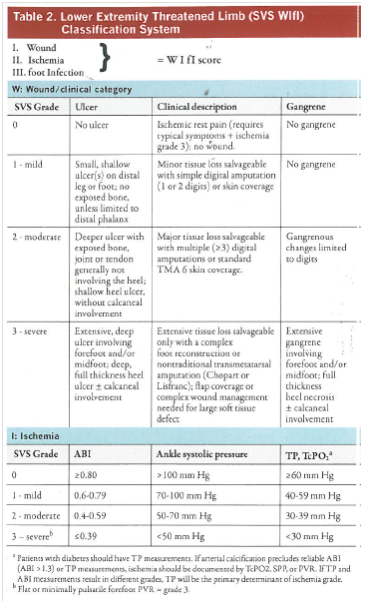
- Prediction of patients most likely to require and to benefit from revascularization can be based on the Society of Vascular Surgery (SVS) Wound, Ischemia, and foot infection (Wifi) lower extremity threatened limb classification. (See Table 2).
- A combination of clinical judgement and careful interpretation of objective assessments of perfusion along with consideration of the wound and infection extent is required to select patients appropriately for revascularization.
- In functional patients with long- segment occlusive disease and a good autologous conduit, bypass is likely to be preferable.
- In the setting of tissue loss and diabetes, prosthetic bypass is inferior to bypass with vein conduit.
- The choice of intervention depends on the degree of ischemia, the extent of arterial disease, the extent of the wound, the presence of absence of infection, and the available expertise.
Treatment
Off-loading DFUs
- In patients with plantar DFU, we recommend off-loading with a total contact cast (TCC) or irremovable fixed ankle cast walker as an alternative to TCC and irremovable fixed ankle walking boot.
- In patients with FDU requiring frequent dressing changes, we suggest off-loading using a removable cast walker as an alternative to TCC and irremovable fixed ankle walking boot.
- We suggest against using postoperative shoes or standard or customary footwear for off-loading plantar DFU’s
- In patients with nonplantar wounds, we recommend using any modality that relieves pressure at the site of the ulcer, such as a surgical sandal or heel relief shoe.
- In high risk patients with healed DFU (including those with a prior history of DFU, partial foot amputation, or Charcot foot. We recommend wearing specific therapeutic foot wear with pressure relieving insoles to aid in prevention of new or recurrent foot ulcers.
Wound Care for DFU’s
- We recommend frequent evaluation at 1-to 4- week intervals with measurements of diabetic foot wounds to monitor reduction of wounds size and healing progress.
- We recommend evaluation for infection on initial presentation of all diabetic foot wound size and healing progress.
- We recommend evaluation for infection on initial presentation of all diabetic foot wounds, with initial sharp debridement of all infected diabetic ulcers, and surgical intervention for foot infections involving abscess, gas, necrotizing fascilitis.
- We suggest that treatment of DFIs should follow the most current guidelines published by the Infectious Diseases Society of America (IDSA)
- We recommend use of dressing products that maintain a moist wound bed, control exudates, and avoid maceration of surrounding intact skin for diabetic wounds.
- We recommend sharp debridement of all devitalized tissue and surrounding callus material from diabetic foot ulceration at 1-to 4- week intervals.
- Considering lack of evidence for superiority of any given debridement technique, we suggest initial sharp debridement with subsequent choice of debridement method based on clinical context, availability of expertise and supplies, patient tolerance and preference, and cost effectiveness.
- For DFUs that fail to demonstrate improvement (>50% wound area reduction) after a minimum of 4 weeks of standard wound therapy, we recommend adjunctive wound therapy options.
- These include negative pressure therapy, biologics (platelet- derived growth factor [PDGF], living cellular therapy, extracellular matrix products, amnionic membrane producr), and hyperbaric oxygen
- Choice of adjuvant therapy is based on clinical findings, availability of therapy, and cost effectiveness. There is no recommendation on ordering of therapy choice.
- Re-evaluation of vascular status, infection control, and off-loading is recommended to ensure optimization before initiation of adjunctive wound therapy
- We suggest the use of negative pressure wound therapy for chronic diabetic foot wounds that do not demonstrate expected healing progression with standard or advanced wound dressings after 4-8 weeks of therapy.
- We suggest consideration of the use of PDGF (becaplermin) for the treatment of DFUs that are recalcitrant to standard therapy.
- We suggest consideration of living cellular therapy using a bilayered keratinocyte/fibroblast construct or a fibroblast – seeded matrix for the treatment of DFUs when recalcitrant to standard therapy.
- We suggest consideration of the use of extracellular matrix products employing acellular human dermis or porcine small intestinal submucosal tissue as an adjunctive therapy for DFU’s when recalcitrant to standard therapy.
- In patients with DFU who have adequate perfusion that fails to respond to 4-6 weeks of conservative management, we suggest hyperbaric oxygen therapy.